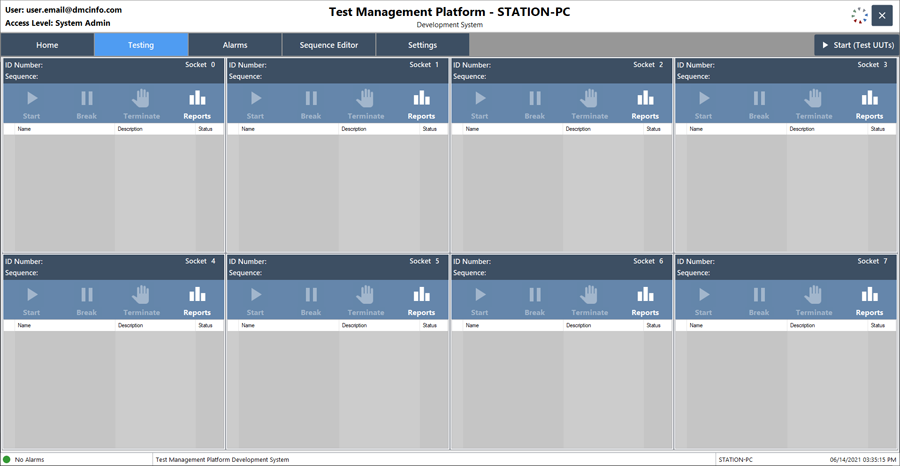
Software Platforms and Validation
With the above requirements in mind, DMC leveraged the NI software platform using both NI TestStand and LabVIEW. TestStand formed the test executive backbone; DMC leveraged the Parallel Process Model to test several devices in parallel, along with the Model Plugin architecture which enabled modular feature design to meet all client requirements. DMC used LabVIEW as the basis of the PC application, acting as the user interface and sending commands to and receiving data from TestStand.
One of the client’s main priorities for the design was validation. Because of our client’s work in the medical device industry, any time a new software version was released, the entire release needed to be revalidated, regardless of the scale of the change. This traditionally led to extended validation efforts with a great deal of paperwork overhead and testing downtime for even the smallest updates.
DMC addressed this concern by allocating core functionality to the TestStand Model Plugins. Model plugins can be added or removed from a station at will without changing anything else about the system configuration. This allows a plug-and-play approach for configuring each station without requiring full software revalidation for each unique test station or plugin configuration. This approach led to reduced validation requirements because each plugin was deployed via an independent installer, meaning that changes to one plugin did not trigger validation needs in any of the other plugins.
Integrating Third-Party Hardware and Software
We chose LabVIEW as the basis for PC application due to the client’s familiarity with the platform and the ease with which new devices and protocols could be integrated into the platform. Our client had proprietary hardware with a communication protocol that was developed in-house. We needed to integrate this protocol and hardware into our system in addition to dozens of different off-the-shelf devices used for testing. Many of the devices provided third-party drivers, and LabVIEW provided the needed tools to create custom drivers for the in-house protocols and hardware.
Balancing Different Needs and Permissions
Our client has multiple products requiring testing with the same unified software, which often creates internal disagreements about software versions and compatibility. Production supports the latest hardware, but the service department needs to maintain older code that is used less often. DMC worked with both departments to support migrating legacy systems to the new platform to standardize all systems on the new platform, while allowing for support of older product models.
Additionally, there was a wide variety of people at the client site who needed to interact with the software for testing. DMC provided configurable permissions integrated directly with the client’s internal LDAP server to allow more in-depth system configuration features for engineers and admin users, while locking down the user interface for operators in production.
Data Tracking
With such a high volume of products and a wide range of data inputs, it was essential for DMC to work with the client on ensuring all servers and tests were running well. DMC added input validation to ensure the system syncs with the server, such as making sure the production server is up to date, and that database communication is live.
Additionally, DMC added a SCADA integration for tracking the type of test for the current asset and whether the unit is approved for the current test. It also uploads results, so the data is centralized in one place and easy to view. This has aided ongoing support to evaluate the reduction of cycle time for key devices and made it easier for the client and DMC to make improvements to the system and the processes over time.
Looking to step up your medical device testing? Check out DMC’s Test & Measurement expertise and contact us for your next project.








